Have we found the true cause of diabetes, stroke, and Alzheimer’s?
Does this one microbe cause heart disease, stroke, Alzheimer’s, diabetes, Parkinson’s, pre-term birth, pancreatic cancer, and kidney disease? Does that mean we can beat them?
For decades, health experts have been lecturing us about our bad habits, blaming them for the surge in “lifestyle diseases.” These often come on as we age and include heart disease, Alzheimer’s, type 2 diabetes, and some cancers. Worldwide, 70 percent of all deaths are now attributed to these conditions. In the U.K., it is a whopping 90 percent.
Too much red meat, too little fruit and veg, smoking, drinking, obesity, and not enough exercise appears to make all these diseases more likely – and having any of them makes getting the others more likely. But no one knows why, and we still haven’t determined what causes them. Alzheimer’s is now one of the U.K.’s biggest killers, yet the main hypothesis for how it originates imploded this year after drugs based on it repeatedly failed. High blood cholesterol is blamed for heart attacks, except most people who have heart attacks don’t have it.
What we do know is that these conditions usually start causing symptoms later in life, and their prevalence is skyrocketing as we live longer. They all turn inflammation, the method our immune system uses to kill invaders, against us. And, by definition, these diseases aren’t infectious. They are down to bad habits and unlucky genes, not germs. Right?
Not necessarily. In disease after disease, we find that bacteria are covertly involved, invading organs, co-opting our immune systems to boost their survival, and slowly making bits of us break down. The implication is that we may eventually be able to defeat heart attacks or Alzheimer’s just by stopping these microbes.
Until now, bacteria’s involvement completely eluded us. They work very slowly, stay dormant for long periods, or hide inside cells. That makes them difficult to grow in culture, once the gold standard for linking bacteria to disease. But now D.N.A. sequencing has revealed bacteria in places they were never supposed to be, manipulating inflammation in the ways observed in these diseases.
The findings are contrary to received wisdom and emerging in so many diseases, each with its separate research community, that awareness of all this is only starting to hit the mainstream (See “Germ theory“). And predictably, as with any paradigm shift, there is resistance.
But some researchers, frustrated by years of failure to find causes, and therefore real treatments, for the diseases of aging, are cautiously excited. And with reason: this could change everything.
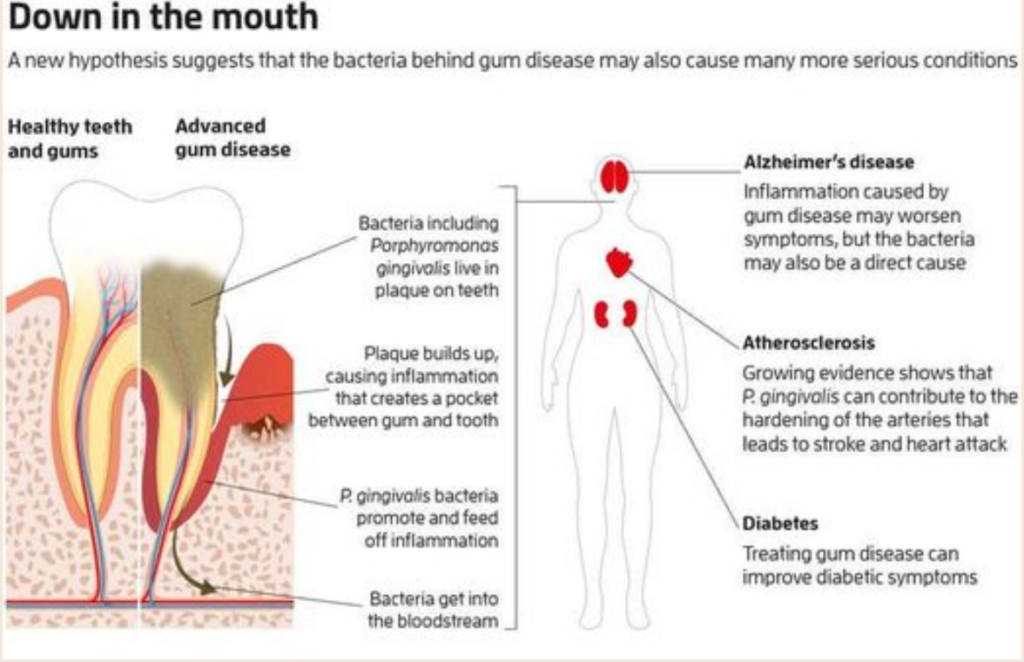
The worst culprits, which play a role in the widest range of ailments, are the bacteria that cause gum disease. This is the most widespread disease of aging – “the most prevalent disease of mankind,” says Maurizio Tonettiat, the University of Hong Kong. In the U.S., 42 percent of those aged 30 or above have gum disease, which rises to 60 percent in those 65 and older. It has been measured at 88 percent in Germany.
Strikingly, many of the afflictions of aging – from rheumatoid arthritis to Parkinson’s – are more likely, more severe, or both in people with gum disease. Some things may go wrong, leading to gum disease and other disorders. However, there is increasing evidence that the relationship is direct: the bacteria behind gum disease help cause the others.
Circumstantial evidence is certainly damning. In the U.S., states that put federal Medicaid funds towards people’s dental costs, including those related to preventing or treating gum disease, ultimately pay between 31 and 67 percent less than states that don’t help those people later with heart attacks, diabetes, strokes, and cancer. David Ojcius at the University of the Pacific in San Francisco says private insurance companies report similar patterns.
But how can the bacteria that cause gum disease play a role in all these conditions? We must look at how they turn the immune system against us to answer that.
Your mouth hosts more than 1000 species of bacteria in a stable community where peaceful bacteria around them keep potential bad actors in check. Elsewhere in the body, including on the skin or the lining of the gut, communities of bacteria live on a continuous sheet of cells, where the outermost layer is constantly shed, getting rid of invasive bacteria. But your teeth can’t cast off a layer like that, says Tonetti. There, the bacteria live on a hard surface, which pierces through the protective outer sheet of cells.
When the plaque the bacteria on your teeth live in builds up enough to harden and spread under the gum, it triggers inflammation: immune cells flood in and destroy both microbes and our infected cells (see Diagram). An oxygen-poor pocket develops between gum and tooth if this goes on too long. A handful of bacteria take advantage of this and multiply. One of them, Porphyromonas gingivalis, is especially insidious, disrupting the stable bacterial community and prolonging inflammation.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7565656/
5.1. Application of Silver Nanoparticles in Dental Medicine
Silver has been used in dentistry for over a century and is a crucial component in dental amalgam fillings [4]. It is used in reconstructive dentistry, as well as in implantology and the production of dentures. Biofilms on the surface of a dental implant can cause inflammatory lesions on the peri-implant mucosa, consequently increasing the risk of implant failure [13]. The main goal of using silver nanoparticles is to prevent infection during and after dental surgery, i.e., thanks to their antibacterial activity, microbial colonization through embedded biomaterials are reduced [4,38,39].
The antimicrobial features of silver nanoparticles have also been studied in detail in dental medicine. Based on the results of the research, there is a growing interest in AgNPs [40]. The oral cavity is an active ecosystem that is regularly colonized by diverse pathogenic microorganisms, so dental materials and implants have an increased risk of infection [13]. In vitro examinations show the unique antimicrobial silver nanoparticles’ action when bound to dental materials such as nanocomposites, acrylic resins, composite resins, adhesives, intracanal drugs and implant coatings [40]. They are also used to make membranes for guided tissue regeneration in periodontal treatment [6]. Smaller silver nanoparticles have increased antibacterial activity against oral anaerobic pathogenic bacteria [6]. It is important to note that AgNPs, thanks to their antitumor properties, have shown positive results in the treatment of oral cancer [40].
The application of silver preparations as a microbicide to stop dentin caries is becoming more common. In vitro experiments demonstrated the microbicidal effectiveness of silver diamine fluoride (SDF) on cariogenic microbes in a human dentin model. Besides, silver nanoparticles have also been recognized in in vitro studies to have a microbicidal impact against growth, adhesion, and biofilm development of Streptococcus mutans in human dentin models. SDF has an intense antimicrobial effect on dental plaque. It reduces the metabolism of carbohydrates in dental plaque and stimulates a different balance of plaque flora [41,42].
SDF has a bactericidal effect on cariogenic bacteria, largely S. mutans, inhibiting the increase in cariogenic biofilms on teeth. Additionally, SDF stimulates remineralization of demineralized enamel or dentin and inhibits collagenases (matrix metalloproteinases and cysteine cathepsins) and thus protects the collagen in dentin from demolition [43].
AgNPs, in combination with antibiotics, enhances bactericidal features. When inactive antibiotics are combined with AgNPs, they gain strong antibacterial activity against multidrug-resistant strains of bacteria [6].
Silver nanoparticles have a better bacteriostatic and bactericidal outcome, with five times lower concentration than chlorhexidine. When AgNPs are used in the proper concentration, it is a safe option than other chemically derived antimicrobials [44]. Dental materials with AgNPs are biocompatible and have no meaningful toxic or mutagenic consequences [6].
IADR/AADR/CADR
IADR Abstract Archives Evaluation of the Antimicrobial Activity of Colloidal Silver in Odontopathogenic Bacteria Objectives: Determine the antimicrobial activity of colloidal silver in three oral bacteria. Methods: Enterococcus faecalis (ATCC 29212), Fusobacterium nucleatum (ATCC 10953) and Streptococcus mutans (ATCC UA159) were obtained from the American Type Culture Collection. The minimal inhibitory concentration (MIC) was performed in 96-well flatbottom plates containing 100μL of Colloidal Silver at 20 ppm in TSB supplemented with 10% FBS and 100 μL of 1.0 × 108 CFU / mL of bacterial strains, up to a final volume of 200μL per well; 5μg / mL chlorhexidine was used as a positive control for inhibition of bacterial growth and saline solution (SS) as a negative control. The plates were incubated at 37 ° C for 72h in a microaerobic atmosphere. Subsequently, the percentage of growth inhibition was calculated using the MIC value. Finally, 10 μL of culture were taken from wells without bacterial strains growth and spread on TSA supplemented with 5% DSB. The CFU were counted after seven days of incubation. The minimum bacterial concentration (MBC) value was established as the low-est concentration of colloidal silver that killed the bacterial strains. Data were evaluated using t-student and ANOVA in SPSS v.21 Results: The data obtained show that the antimicrobial activity of the colloidal silver of 20 ppm was 99.9% and therefore showed no microbial growth for the bacterial strains evaluated compared to the control group of chlorhexidine. A statistically significant difference was observed (p< 0.05) between the groups evaluated. Conclusions: Colloidal silver at a concentration of 20 ppm has antimicrobial activity against odontopathogenic bacteria and could therefore be used as an antimicrobial in the oral cavity. Division: IADR/AADR/CA
https://www.frontiersin.org/articles/10.3389/fcimb.2022.987683/full

Colloidal Silver As ions (Ag+) have been recognized for their antimicrobial properties, which include the ability to prevent implant-associated infections by anaerobic bacteria in vitro and in vivo. An ionic silver coating exhibited antimicrobial activity against anaerobic bacteria like P. gingivalis, indicating its potential for preventing infection in implant materials. Specifically, silver ions have been used to treat P. gingivalis-inoculated agar gels, demonstrating their efficacy against anaerobic bacteria.
The interaction between Porphyromonas gingivalis and silver ions is particularly interesting due to the potential implications for managing and preventing the spread of this bacterium. Research has shown that silver ions can inhibit the growth and activity of P. gingivalis, suggesting a possible role for silver ions in treating and preventing periodontitis and associated systemic diseases. However, further research is needed to fully understand the mechanisms of action and develop effective strategies for utilizing silver ions in managing P. gingivalis-related conditions.
Your mouth hosts more than 1000 species of bacteria in a stable community where peaceful bacteria around them keep potential bad actors in check. Elsewhere in the body, including on the skin or the lining of the gut, communities of bacteria live on a continuous sheet of cells, where the outermost layer is constantly shed, getting rid of invasive bacteria. But your teeth can’t cast off a layer like that, says Tonetti. There, the bacteria live on a hard surface, which pierces through the protective outer sheet of cells.
When the plaque the bacteria on your teeth live in builds up enough to harden and spread under the gum, it triggers inflammation: immune cells flood in and destroy both microbes and our infected cells. If this goes on too long, an oxygen-poor pocket develops between gum and tooth. A handful of bacteria take advantage of this and multiply. One of them, Porphyromonas gingivalis, is especially insidious, disrupting the stable bacterial community and prolonging inflammation.
This is a strange thing to do. Most pathogens try to block or avoid inflammation, which normally kills them before it shuts down again. Starting in our 30s and 40s, this shutdown begins failing, leading to the chronic inflammation involved in diseases of aging. No one knows why.
P. gingivalis may have a hand in it. It perpetuates inflammation by producing molecules that block some inflammatory processes, but not all of them, says Caroline Genco of Tufts University in Massachusetts. The resulting weakened inflammation never quite destroys the bacteria but keeps trying, killing your cells. The debris is a feast for P. gingivalis, which, unlike most bacteria, needs to eat protein.
They are left that that that that that that that that that that that that that that that The destruction also liberates the iron that bacteria need and which the body, therefore, normally keeps locked up. “These bacteria manipulate their interaction with the host immune response to enhance their survival,” says George Hajishengallis at the University of Pennsylvania.
[link to theartofmakingcolloidalsilver.com (secure)]
A friend went through this, and this is what he did:
He took colloidal Silver in, washed his mouth with it, swished it, and saw the inflammation go down. Again, he saw pus trying to come out. When it did, he just pressed his inflamed gum. He pushed it to force the pus out when it was ready to come out until blood had a chance to run and white blood cells could start working on the infection. Then he swished and rinsed with colloidal Silver until no more blood came out. He continued to use colloidal Silver until his mouth was normal, only about 12 hours later.