Millions are dying from drug-resistant infections, global report says.
Antibiotics may no longer work because the bacteria they are intended to kill is becoming resistant.
More than 1.2 million people died worldwide in 2019 from infections caused by bacteria resistant to antibiotics, according to the largest study of the issue to date.
This is more than the annual death toll from malaria or Aids.
Poorer countries are worst affected but antimicrobial resistance threatens everyone’s health, the report says.
Urgent investment in new drugs and using current ones more wisely are recommended to protect against it.
The overuse of antibiotics in recent years for trivial infections means they are becoming less effective against serious infections.
People are dying from common, previously treatable infections because the bacteria that cause them have become resistant to treatment.
UK health officials recently warned antimicrobial resistance (AMR) was a “hidden pandemic” that could emerge in the wake of Covid-19 unless antibiotics were prescribed responsibly.
Particularly deadly
The estimate of global deaths from AMR, published in the Lancet, is based on an analysis of 204 countries by a team of international researchers, led by the University of Washington, US.
They calculate up to five million people died in 2019 from illnesses in which AMR played a role – on top of the 1.2 million deaths it caused directly.
In the same year, Aids (acquired immune deficiency syndrome) is thought to have caused 860,000 deaths and malaria 640,000.
Most of the deaths from AMR were caused by lower respiratory infections, such as pneumonia, and bloodstream infections, which can lead to sepsis.
MRSA (methicillin-resistant Staphylococcus aureus) was particularly deadly, while E. coli, and several other bacteria, were also linked to high levels of drug resistance.
Using patient records from hospitals, studies and other data sources, the researchers say young children are at most risk, with about one in five deaths linked to AMR being among the under-fives.
Deaths from AMR were estimated to be:
- highest in sub-Saharan Africa and South Asia, at 24 deaths in every 100,000
- lowest in high-income countries, at 13 in every 100,000
Prof Chris Murray, from the Institute for Health Metrics and Evaluation at the University of Washington, said the new data revealed the true scale of antimicrobial resistance worldwide and was a clear signal immediate action was needed “if we want to stay ahead in the race against antimicrobial resistance.
Other experts say better tracking of resistance levels in different countries and regions is essential.
Dr Ramanan Laxminarayan, from the Centre for Disease Dynamics, Economics and Policy, in Washington DC, said global spending on addressing AMR needed to rise to levels seen for other diseases.
“Spending needs to be directed to preventing infections in the first place, making sure existing antibiotics are used appropriately and judiciously, and to bringing new antibiotics to market,” he said.
Much of the world faced the challenge of poor access to affordable, effective antibiotics – and that needed to be taken seriously by political and health leaders everywhere, Dr Laxminarayan added.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2292600/?report=classic
Antibacterial Activity and Mechanism of Action of the Silver Ion in Staphylococcus aureus and Escherichia coli▿
The antibacterial effect and mechanism of action of a silver ion solution that was electrically generated were investigated for Staphylococcus aureus and Escherichia coli by analyzing the growth, morphology, and ultrastructure of the bacterial cells following treatment with the silver ion solution. Bacteria were exposed to the silver ion solution for various lengths of time, and the antibacterial effect of the solution was tested using the conventional plate count method and flow cytometric (FC) analysis. Reductions of more than 5 log10 CFU/ml of both S. aureus and E. coli bacteria were confirmed after 90 min of treatment with the silver ion solution. Significant reduction of S. aureus and E. coli cells was also observed by FC analysis; however, the reduction rate determined by FC analysis was less than that determined by the conventional plate count method. These differences may be attributed to the presence of bacteria in an active but nonculturable (ABNC) state after treatment with the silver ion solution. Transmission electron microscopy showed considerable changes in the bacterial cell membranes upon silver ion treatment, which might be the cause or consequence of cell death. In conclusion, the results of the present study suggest that silver ions may cause S. aureus and E. coli bacteria to reach an ABNC state and eventually die.
https://pubmed.ncbi.nlm.nih.gov/11033548/
To investigate the mechanism of inhibition of silver ions on microorganisms, two strains of bacteria, namely Gram-negative Escherichia coli (E. coli) and Gram-positive Staphylococcus aureus (S. aureus), were treated with AgNO(3) and studied using combined electron microscopy and X-ray microanalysis. Similar morphological changes occurred in both E. coli and S. aureus cells after Ag(+) treatment. The cytoplasm membrane detached from the cell wall. A remarkable electron-light region appeared in the center of the cells, which contained condensed deoxyribonucleic acid (DNA) molecules. There are many small electron-dense granules either surrounding the cell wall or depositing inside the cells. The existence of elements of silver and sulfur in the electron-dense granules and cytoplasm detected by X-ray microanalysis suggested the antibacterial mechanism of silver: DNA lost its replication ability and the protein became inactivated after Ag(+) treatment. The slighter morphological changes of S. aureus compared with E. coli recommended a defense system of S. aureus against the inhibitory effects of Ag(+) ions.
Leader of global antibiotic research accelerator discusses urgent need for innovations to combat superbugs
The federal government recently reinforced the importance of the fight against superbugs when it released an updated National Action Plan for Combating Antibiotic-Resistant Bacteria, but the guidance to agencies lacks details on specific policies and on ways to achieve key priorities.
The October update of the plan first published in 2015 calls for continued work in many essential areas, identified earlier by Pew and others, to fix the broken antibiotics market and promote antibiotic stewardship. The document highlights the need to implement policies that stimulate development of urgently needed drugs, strengthen the country’s capacity to track antibiotic resistance and use, and improve antibiotic use in humans and animals. Still, the 2020 update would have benefited from more specifics on the economics of essential antibiotic development and what steps should be taken.
For example, the new action plan lacks a mechanism to ensure measurable progress in the implementation of a package of economic incentives to boost investment in antibiotic discovery and development and to create a sustainable market for these lifesaving drugs. To help jump-start the development of new antibiotics, lawmakers have already introduced bipartisan measures, such as the Pioneering Antimicrobial Subscriptions To End Up Surging Resistance (PASTEUR) Act of 2020 and the Developing an Innovative Strategy for Antimicrobial Resistant Microorganisms (DISARM) Act of 2019. The recent update, however, includes no mention of such approaches.
The plan does call for the continuation of existing government efforts to stimulate antibiotic innovation, including adjustments by the Centers for Medicare & Medicaid Services to antibiotic reimbursement as well as initiatives from the Biomedical Advanced Research and Development Authority to purchase antibiotics for national security purposes. However, these efforts are not sufficient to repair the antibiotic market.
Much like the 2015 national action plan, the update focuses on supporting antibiotic discovery and development rather than enacting incentives to adequately address the troubled market. Additionally, the new plan does not mention the need to improve the sharing of antibiotic research data, which experts agree is essential for catalyzing urgently needed antibiotic discovery.
In addition, the government and health care stakeholders must improve the capacity to monitor antibiotic use and resistance so they can quickly identify and combat superbug outbreaks. Effective stewardship policies for reducing inappropriate prescribing also are essential. Increasing the number of hospitals reporting antibiotic use and resistance data to the Centers for Disease Control and Prevention’s National Healthcare Safety Network would represent fundamental progress in this arena. The 2020 plan sets a series of goals for hospital reporting but lacks specific strategies and policies to reach those goals by 2025.
Any national strategy should also promote appropriate antibiotic use in humans and food-producing animals to combat antibiotic-resistant bacteria. Research shows that the more often antibiotics are used, the less effective they become. However, the updated plan falls short on both fronts.
In antibiotic use in human medicine, Pew strongly supports the prioritization of improving antibiotic prescribing in doctor’s offices and other outpatient health care settings. Research from CDC and Pew shows that 1 in 3 outpatient antibiotic prescriptions is unnecessary, which puts patients needlessly at risk and accelerates the emergence of resistance. Although the 2020 national action plan calls for a decrease in antibiotic prescribing in outpatient settings and improved timelines for outpatient antibiotic use reporting, it does not include specific targets or interventions for developing antibiotic stewardship in these settings. Such programs help to ensure that these drugs are used appropriately and only when necessary.
Regarding the judicious antibiotic use in farm animals, the plan includes no direct reference to the Food and Drug Administration’s five-year plan to improve antibiotic stewardship in these animals. It also lacks specifics on how and when the agency will collect and publicly share important new information about antibiotic use in animals. For example, there is no mention of when or how FDA will start adjusting antibiotic sales data based on the size of animal populations—a step that that would provide valuable context to the meager existing information on agricultural antibiotic use. The agency has committed to doing this but has yet to take any concrete action.
In the same vein, the plan does not include specifics or timelines for establishing evidence-based durations of use for all medically important antibiotics given to the animals and other steps to ensure that these drugs are used judiciously. Currently, many antibiotics can be given to animals for weeks, months, or even indefinitely. Failure to address this problem in the new action plan is a clear omission.
The updated plan signals that the fight against antibiotic resistance is—and should remain—a national priority, but a lack of clear benchmarks, metrics, and strategies for achieving its stated goals is a missed opportunity. Pew encourages federal agencies to take steps to improve this and future national action plans and looks forward to coordinating with policymakers and other stakeholders to drive meaningful progress on the real and growing threat posed by superbugs.
With the world’s attention on COVID-19, I believe that now is the time to talk about another pandemic that’s been happening right under our noses: antimicrobial resistance (AMR).
When infections caused by bacteria, parasites, viruses or fungi stop responding to the medicines designed to treat them, that’s AMR. Resistance builds over time through overexposure to antimicrobial drugs, such as antibiotics, or disinfectants. With ineffective treatments, these infections persist in the body and ultimately spread to others.
AMR is a slower-moving pandemic than COVID-19, but one that is worsening every day. A recent report by the Council of Canadian Academies said that in 2018, more than a quarter of all infections in Canada were resistant to first-line drugs. In that one year alone, 5,400 people died as a direct result of resistant infections.
Drug-resistant infections lead to longer hospital stays and potentially greater complications. Doctors might need to use alternative medicines with more side effects. These issues cost the Canadian health-care system $1.4 billion, and this will only increase.
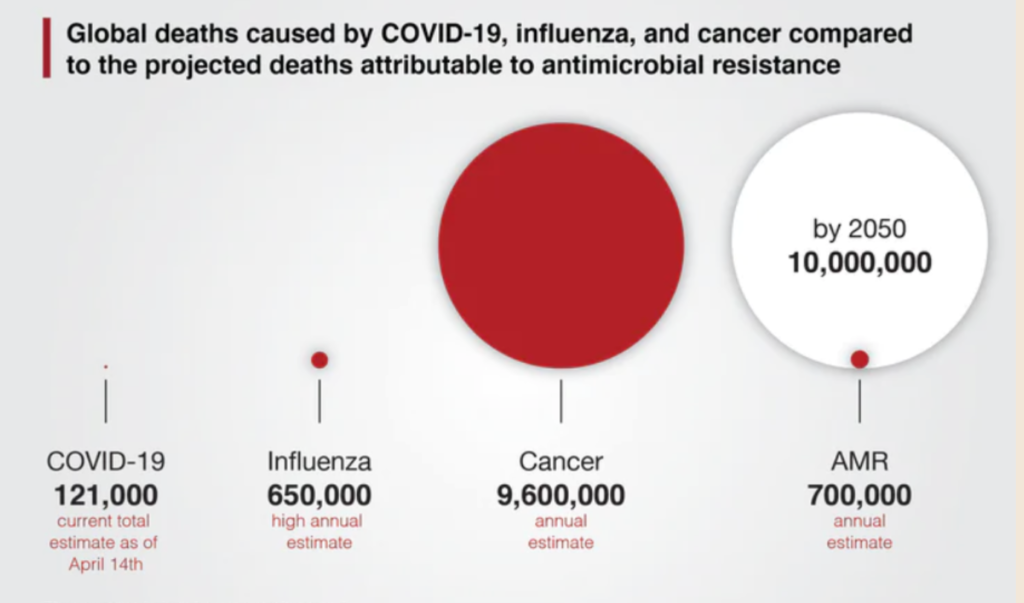
The United Nations, the World Health Organization and even the World Bank recognize AMR as a public health and economic disaster waiting to happen. In 2016, United Kingdom economist Jim O’Neill and his team published a report stating that if we do not address the growing threat of AMR, by 2050 more people will die from drug-resistant infections than from cancer.
If left unsolved, AMR will cause more global deaths than cancer by 2050. Graphic: Christy Groves, Michael G. DeGroote Institute for Infectious Disease Research, McMaster University.
If left unsolved, AMR will cost the health-care system trillions of dollars and, more importantly, it will cost millions of people their lives.
We can learn from COVID-19. The current pandemic shows that despite all of our medical advances, we remain incredibly vulnerable to infections for which we have no therapies. However, it shows that if sufficiently motivated, we can make huge changes in short timeframes.
While there is inspiring work being done in different parts of the world to address the issues surrounding AMR — including here at McMaster University’s Institute for Infectious Disease Research, where my group is based — there is a long road ahead of us.
The age of antibiotics is coming to an end. When will some brave soul in the medical establishment try silver ions as an alternative? The continued overuse of antibiotics has virtually assured the eventual emergence of a lethal airborne superbug.
“In a bid to battle antibiotic resistance, the World Health Organization for the first time has classified antibiotics into three categories, including one that lists the drugs it hopes will not be used except in circumstances of dire need. The antibiotic breakdown is part of the WHO’s essential medicines list, which is updated every two years. The 2017 iteration was published Tuesday.
The section on antibiotics divides these critical drugs into three categories. Those that should be go-to drugs for common infections are listed as “access” antibiotics. Others for which growing resistance is a problem are listed under the category “watch” to signal they should not be first or second options.
The final category is “reserve” — a name that signals that these are drugs that should only be used to treat infections for which other options have failed.
“These medicines should be protected and prioritized as key targets in [antibiotic] stewardship programs to preserve their effectiveness,” Dr. Marie-Paule Kieny, the WHO’s assistant director-general for health systems and innovation, said during a news conference.
Kieny said the hope is that the list will help health systems and doctors use antibiotics to better preserve their usefulness. For instance, the drug ciprofloxacin shouldn’t be prescribed for urinary tract or upper respiratory tract infections — even though it often is.
“We don’t see this as a quick fix solution,” Kieny said of the ranking tool. “But we know that where preservation of antibiotics has worked, through hospital stewardship programs, we have seen a reduction in resistance trends.”
Outside experts hailed the decision to rank antibiotics in this way.
“The updated essential medicines list should help ensure antibiotics of last resort are reserved only for when first-line treatments will not work and that doctors in all countries have easy access to the best and most appropriate treatments,” Dr. Tim Jinks, head of the Wellcome Trust’s drug-resistant infection program, said in a statement.
Among the drugs in the reserve category is colistin, an old antibiotic that has been a drug of last resort. However, concern about its continued usefulness has arisen because of the emergence of a superbug called mcr-1 — a gene that allows bacteria to withstand the drug.
Dr. Suzanne Hill, WHO’s director of essential medicines and health products, said having a list of antibiotics the world shouldn’t use unless necessary will require a rethinking of how manufacturers are compensated for making drugs in that class.
“We don’t want colistin used very frequently. We don’t want it used at all,” she said.
Pharmaceutical companies typically recoup their development costs through sales, but in these drugs, the goal is to restrict sales to the most significant degree possible. Other forms of compensation — ways to reward companies for not promoting use — might include cash awards or buying out licenses, Hill said.”
From WIRED
WHEN ALEXANDER FLEMING came back from a Scottish vacation in the summer of 1928 to find his London lab bench contaminated with a mold called Penicillium notatum, he kicked off a new age of scientific sovereignty over nature. Since then, the antibiotics he discovered and the many more he inspired have saved millions of lives and spared immeasurable suffering around the globe. But from the moment it started, scientists knew the age of antibiotics came stamped with an expiration date. They just didn’t know when it was.
Bacterial resistance to antibiotics is both natural and inevitable. By the luck of the draw, a few bacteria will have genes that protect them from drugs, and they’ll pass those genes around—not just to their progeny, but sometimes to their neighbors too. Now, computational epidemiologists are finally getting the data and processing to model that phenomenon. But no one’s using these tools to predict the end of the antibiotic era—because it’s already here. Instead, they’re focusing their efforts on understanding how soon resistant bacteria could be in the majority, and what, if anything, doctors can do to stop them.
In 2013, then-director of the Centers for Disease Control and Prevention Tom Frieden told reporters, “If we’re not careful, we will soon be in a post-antibiotic era.” Today, just four years later, the agency says we’ve arrived. “We say that because pan-resistant bacteria are now here” says Jean Patel, who leads the CDC’s Antibiotic Strategy & Coordination Unit. “Folks are dying simply because there is no antibiotic available to treat their infection, infections that not too long ago were easily treatable.”
Last August, a woman in her 70s checked into a hospital in Reno, Nevada with a bacterial infection in her hip. The bug belonged to a class of particularly tenacious microbes known as carpabenem-resistant Enterobacteriaceae, or CREs. Except in addition to carpabenem, this bug was also resistant to tetracycline, and colistin, and every single other antimicrobial on the market, all 26 of them. A few weeks later she developed septic shock and died.
For public health officials like Patel, that case marks the end of an era, and the beginning of a new one. Now, the question is: How fast is that kind of pan-resistance going to spread? “When does it get to the point where it’s more common to have an infection that can’t be treated with antibiotics than one that can?” says Patel. “That’s going to be a very hard thing to predict.”
She knows because she’s tried before. Back in 2002, the first vancomycin-resistant staph infection showed up in a 40-year old Michigan man with a chronic foot ulcer. That seemed really bad: Staph is one of the most common bacterial infections in humans, and vancomycin its most common antibiotic adversary. Plus, the resistance gene was located on a plasmid—a free-floating circle of DNA that makes it easy to get around. Epidemiologists at the CDC worked with microbiologists like Patel to build a model to predict how far and how fast it would spread. While Patel couldn’t remember the exact output, she recalls that the results were scary. “We were very, very concerned about this,” she says.
Luckily in this case, their models were completely mistaken. Since 2002 there have been only 13 cases of vancomycin-resistant staph, and no one has died.
Being so wrong baffled the teams. But biology can be complicated like that. “I’ve worked with these bacteria in labs where they grow just fine, but they don’t seem to spread from one person to another,” says Patel. And while they still don’t know why, one hypothesis is that these particular resistance genes came with a cost. They might have made the staph capable of standing up to its antibiotic archnemesis, but the same bits of DNA also might have made it harder to survive outside a human body. Hospital protocols, time of year, and geography could have also had an effect on transmission rates. It’s more like trying to predict the weather than anything else.
From WIRED
PEER INTO THE POST-APOCALYPTIC FUTURE OF ANTIMICROBIAL RESISTANCE
ABOUT 4 MILLION years ago, a cave was forming in the Delaware Basin of what is now Carlsbad Caverns National Park in New Mexico. From that time on, Lechuguilla Cave remained untouched by humans or animals until its discovery in 1986—an isolated, pristine primeval ecosystem.
When the bacteria found on the walls of Lechuguilla were analyzed, many of the microbes were determined not only to have resistance to natural antibiotics like penicillin, but also to synthetic antibiotics that did not exist on earth until the second half of the twentieth century. As infectious disease specialist Brad Spellberg put it in the New England Journal of Medicine, “These results underscore a critical reality: antibiotic resistance already exists, widely disseminated in nature, to drugs we have not yet invented.”
The origin story of antibiotics is well known, almost mythic, and antibiotics, along with the other basic public health measures, have had a dramatic impact on the quality and longevity of our modern life. When ordinary people called penicillin and sulfa drugs miraculous, they were not exaggerating. These discoveries ushered in the age of antibiotics, and medical science assumed a lifesaving capability previously unknown.

LITTLE, BROWN
Note that we use the word discoveries rather than inventions. Antibiotics were around many millions of years before we were. Since the beginning of time, microbes have been competing with other microbes for nutrients and a place to call home. Under this evolutionary stress, beneficial mutations occurred in the “lucky” and successful ones that resulted in the production of chemicals—antibiotics—to inhibit other species of microbes from thriving and reproducing, while not compromising their own survival. Antibiotics are, in fact, a natural resource—or perhaps more accurately, a natural phenomenon—that can be cherished or squandered like any other gift of nature, such as clean and adequate supplies of water and clean air.
Equally natural, as Lechuguilla Cave reminds us, is the phenomenon of antibiotic resistance. Microbes move in the direction of resistance in order to survive. And that movement, increasingly, threatens our survival.
With each passing year, we lose a percentage of our antibiotic firepower. In a very real sense, we confront the possibility of revisiting the Dark Age where many infections we now consider routine could cause severe illness, when pneumonia or a stomach bug could be a death sentence, when a leading cause of mortality in the United States was tuberculosis.
The Review on Antimicrobial Resistance (AMR) determined that, left unchecked, in the next 35 years antimicrobial resistance could kill 300,000,000 people worldwide and stunt global economic output by $100 trillion. There are no other diseases we currently know of except pandemic influenza that could make that claim. In fact, if the current trend is not altered, antimicrobial resistance could become the world’s single greatest killer, surpassing heart disease or cancer.
In some parts of the United States, about 40 percent of the strains of Streptococcus pneumonia, which the legendary nineteenth and early twentieth century physician Sir William Osler called “the captain of the men of death,” are now resistant to penicillin. And the economic incentives for pharmaceutical companies to develop new antibiotics are not much brighter than those for developing new vaccines. Like vaccines, they are used only occasionally, not every day; they have to compete with older, extremely cheap generic versions manufactured overseas; and to remain effective, their use has to be restricted rather than promoted.
As it is, according to the CDC, each year in the United States at least 2,000,000 people become infected with antibiotic-resistant bacteria and at least 23,000 people die as a direct result of these infections. More people die each year in this country from MRSA (methicillin-resistant Staphylococcus aureus, often picked up in hospitals) than from AIDS.
If we can’t—or don’t—stop the march of resistance and come out into the sunlight, what will a post-antibiotic era look like? What will it actually mean to return to the darkness of the cave?
Without effective and nontoxic antibiotics to control infection, any surgery becomes inherently dangerous, so all but the most critical, lifesaving procedures therefore would be complex risk-benefit decisions. You’d have a hard time doing open-heart surgery, organ transplants, or joint replacements, and there would be no more in vitro fertilization. Caesarian delivery would be far more risky. Cancer chemotherapy would take a giant step backwards, as would neonatal and regular intensive care. For that matter, no one would go into a hospital unless they absolutely had to because of all the germs on floors and other surfaces and floating around in the air. Rheumatic fever would have lifelong consequences. TB sanitaria could be back in business. You could just about do a post-apocalyptic sci-fi movie on the subject.
To understand why antibiotic resistance is rapidly increasing and what we need to do to avert this bleak future and reduce its impact, we have to understand the Big Picture of how it happens, where it happens, and how it’s driven by use in humans and animals.
Human Use
Think of an American couple, both of who work fulltime. One day, their 4-year-old son wakes up crying with an earache. Either mom or dad takes the child to the pediatrician, who has probably seen a raft of these earaches lately and is pretty sure it’s a viral infection. There is no effective antiviral drug available to treat the ear infection. Using an antibiotic in this situation only exposes other bacteria that the child may be carrying to the drug and increases the likelihood that an antibiotic resistant strain of bacteria will win the evolutionary lottery. But the parent knows that unless the child has been given a prescription for something, the daycare center isn’t going to take him and neither partner can take off from work. It doesn’t seem like a big deal to write an antibiotic prescription to solve this couple’s dilemma, even if the odds the antibiotic is really called for are minute.
While the majority of people understand that antibiotics are overprescribed and therefore subject to mounting resistance, they think the resistance applies to them, rather than the microbes. They believe that if they take too many antibiotics – whatever that unknown number might be—they will become resistant to the agents, so if they are promoting a risk factor, it is only for themselves rather than for the entire community.
Doctors, of course, understand the real risk. Are they culpable to the charge of over- and inappropriately prescribing antibiotics? In too many cases, the answer is Yes.
Why do doctors overprescribe? Is it about covering their backsides in this litigious society? Is it a lack of awareness of the problem? According to Brad Spellberg, “The majority of the problem really revolves around fear. It’s not any more complicated than that. It’s brain stem level, sub-telencephalonic, not-conscious-thought fear of being wrong. Because we don’t know what our patients have when they’re first in front of us. We really cannot distinguish viral from bacterial infections. We just can’t.”
Spellberg cited a case, one he heard at an infectious disease conference he attended. A 25-year-old woman came into the urgent care facility of a prominent health care network complaining of fever, sore throat, headache, runny nose and malaise. These are the symptoms of a classic viral syndrome and the facility followed exactly the proper procedure. They didn’t prescribe an antibiotic, but instead told her to go home, rest, keep herself hydrated, maybe have some chicken soup, and they would call her in three days to make sure she was all right.
She came back a week later in septic shock and died soon after.
“It turns out she had Lemierre’s disease,” says Spellberg. “It clotted her jugular vein from a bacterial infection that spread from her throat to her bloodstream. This is about a one-in-10,000 event; it’s pretty darn rare. But it’s a complication of an antecedent viral infection, and it’s a known complication. So this patient, ironically, would have benefitted from receiving inappropriate antibiotics. How many times do you think doctors need to have those things happen before they start giving antibiotics to every person who walks in the door?”
As much difficulty as we’re having controlling antibiotic resistance in the First World, for the rest, we believe the situation to be a whole lot worse.
In many of these countries, antibiotics are sold right over the counter just like aspirin and nasal spray; you don’t even need a doctor’s prescription. While we in the public health community would certainly like to see a complete cessation of antibiotic use without a prescription, how do we tell sick people in developing countries that they first have to see a doctor, when there may be no more than one or two physicians for thousands of individuals, and even if they could find one, they couldn’t afford the visit in the first place? Taking an action in a vacuum, such as banning over-the-counter sales without improving infrastructure, simply isn’t viable.
The medical community is once again stressing out at the renewed emergence of gram-negative superbugs. Silver ions are proven to be very effective at killing gram-negative bacteria of all types. The medical community would seem to prefer to keep pumping more and more antibiotics into people, therefore, ensuring that even more deadly strains of these bacteria emerge. They seem to want to wait for a vaccine which some pharmaceutical company can then sell. Why not try 50 ppm ionic silver.
A dangerous strain of antibiotic-resistant bacteria is far more common in Houston than anyone knew and shows signs it can spread prolifically, researchers reported [Tue 16 May 2017].
It’s a specific strain of bacteria known as _Klebsiella pneumoniae_. The superbug is showing a special talent for picking up genes that give it the ability to resist a broad range of antibiotics, the team at Houston Methodist hospital system said. “It causes very serious
infection in hospitalized patients,” said Dr James Musser, who led the study team. “What we discovered, surprisingly, is one of the major strains causing infection in our patients in Houston is genetically distinct from strains of this same germ causing human infections
[elsewhere] in the United States,” Musser added.
Doctors and public health officials have been warning for years about the rise of drug-resistant superbugs. They are especially dangerous to patients in the hospital for extended periods of time but they can also be found in people with ordinary infections such as cystitis and pneumonia.
There have been some nightmarish outbreaks — like one in 2011 at the flagship National Institutes of Health’s clinical center in Bethesda, Maryland, that killed 7 people. That outbreak was caused by a strain of _Klebsiella_ resistant to a major class of antibiotics called carbapenems. Officials eventually discovered it was breeding in the hospital’s sinks. And just this past January [2017], doctors reported on the case of a Nevada woman who had traveled to India and died from a rare superbug that could not be killed by any antibiotic available in the U.S.
These preliminaries settled, he did not care to put off any longer the execution of his design, urged on to it by the thought of all the world was losing by his delay, seeing what wrongs he intended to right, grievances to redress, injustices to repair, abuses to remove, and duties to discharge.
Date: Wed 17 May 2017 3:58 PM ET
Source: NBC News [edited]
<http://www.nbcnews.com/health/health-news/houston-s-had-superbug-problem-years-never-knew-n760466>
To observe how the bacterium Escherichia coli adapted to increasingly higher doses of antibiotics, researchers divided the dish into sections and saturated them with various doses of medication. The outermost rims of the dish were free of any drug. The next section contained a small amount of antibiotic — just above the minimum needed to kill the bacteria — and each subsequent section represented a 10-fold increase in dose, with the center of the dish containing 1,000 times as much antibiotic as the area with the lowest dose.
Over two weeks, a camera mounted on the ceiling above the dish took periodic snapshots that the researchers spliced into a time-lapsed montage. The result? A powerful, unvarnished visualization of bacterial movement, death, and survival; evolution at work, visible to the naked eye.
#282
Excellent video! I just sent the message below to Harvard Medical School:
Greetings! The link above has a neat video showing increase in bacteria resistance through higher concentrations of antibiotics. I would like to see a similar test, but including a strip along the side containing colloidal silver with concentrations increasing from PPB to PPM. My expectation is that low levels of colloidal silver will prove to be more effective than high concentrations of antibiotics.
#283
Excellent weatherman!
On the off chance that they reply, you can tell them that I will be happy to donate an ultra for the project. They can generate a half gallon of 50 ppm and titrate the values down from there.
#284They won’t respond to weatherman so do not hold your breath abe.
They are puppets to the money kings in Big Pharma.
The last thing they want to do is supply evidence EIS / CS works effectively over the man-made concoctions.
Idiots with money paying idiots with a technology ability and NEITHER have ETHICS.
Jul 14, 2020#849
Viruses cause most upper respiratory tract infections (URTIs), such as adenovirus, coronavirus, coxsackievirus, influenza virus, parainfluenza virus, respiratory syncytial virus, and rhinovirus, which account for the majority of cases. (1) A broad-spectrum anti-viral agent that really works is needed to combat over 200 viruses that cause URTIs. (2) Undoubtedly oligodynamic silver fits this bill.
Emerging medical studies confirm the stellar, broad-spectrum virotoxic efficacy of oligodynamic silver both in vitro and in vivo. This includes some of the most formidable viral organisms like HIV (including co-infections) (3-11) and Herpesvirus hominis (HSV). (12-18) Despite the low yields of oligodynamic silver of the past 100 years common to silver-based drugs, the collective authoritative medical literature has documented efficacy of silver’s virotoxicity against over 24 viruses. For the viruses relevant to URTIs, the following are known to succumb to oligodynamic silver:
Oligodynamic silver’s antimicrobial efficacy extends well beyond its virotoxicity. Oligodynamic silver’s lethal effects span across all microbial domains (viral, bacterial, and fungal). The following URTI-related bacteria are known to be susceptible to oligodynamic silver:
Beta hemolytic streptococci, which causes tonsillopharyngeal cellulitis, tonsillopharyngeal abscess (including reduced nasopharangeal abscesses), otitis media, plus sinusitis, and up to ten percent of cases of adult pharyngitis and the associated condition, and scarlet fever; Bordetella pertussis; (causes less than 10% of acute tracheobronchitis cases); Spring Catarrh (obsolete nomenclature); and Pneumococci /Pneumonia; And finally, inflammatory conditions of the eyes, ears, nose, and throat. Streptococcus pneumoniae; Corynebacterium diphtheriae; Neisseria gonorrhoeae; Herpesvirus hominis ; Klebsiella pneumoniae; Haemophilus influenzae; Mycobacterium (Tuberculosis);
Case History
Perhaps oligodynamic silver’s most compelling nature lies in its ability to successfully eradicate pervasive primary and secondary co-infections simultaneously. A major, double-blind, controlled trial concerning advanced AIDS candidiasis and immunity-suppressing moieties demonstrated complete sero-negative conversion after a single treatment with oligodynamic silver hydrosol! The studies were conducted at Lucha Contra el Sida, Comayaguela, Tegucigalpa, Honduras, Central America.
Quoting from the study, “Furthermore, said devices [silver oxide hydrosol] are capable of killing pathogens and purging the bloodstream of immune suppressing moieties (ISM) whether or not created by the AIDS virus (HIV), so as to restore the immune system.” (86) (Brackets added by authors.)
This single treatment delivered a total of 200 mg of silver for a 70 kilogram patient, well within the lowest observed adverse event level (LOAEL) established by the EPA for injected silver. (87) Unlike picoscalar oligodynamic silver hydrosol devoid of silver oxide, the former required activation into an oligodynamic state with persulfate. Nevertheless, the results were astounding.
Two new studies out this week highlight the growing danger of antibiotic-resistant bacteria to global health.
The most alarming finding was that one type of dangerous bacteria could be spreading more rapidly—and mutating faster—than previously recognized, according to a study published in the Proceedings of the National Academy of Sciences on Jan. 16.
What Is a Superbug?
Every year, over two million people in the U.S. get infections that are resistant to some types of antibiotics. On the ‘Consumer 101‘ TV show, Consumer Reports’ expert Lauren Friedman explains what you need to know about these superbug infections.
The bacteria in question is called “carbapenem-resistant Enterobacteriaceae,” or CRE for short, and can cause serious infections throughout the body, including in the lungs, bladder, bloodstream, and skin.
The infections, which mostly occur in medical facilities, are often hard to treat—or are even untreatable—because the bacteria are resistant to many antibiotics.
Just last week, the Centers for Disease Control and Prevention reported that a Nevada woman in her 70s had died after being infected with a “nightmare bacteria” (a type of CRE) resistant to all antibiotics.
Then yesterday, the New England Journal of Medicine published findings from research conducted in South Africa showing that a strain of tuberculosis immune to most antibiotics spreads more easily than previously thought. That’s concerning because the antibiotic-resistant form of tuberculosis—which is rare in the U.S.—though deadly, was not previously considered highly contagious.
Although the deadly strain of tuberculosis has not yet made it to the U.S., the spread of the disease in South Africa is an important reminder that we need to take precautions to contain CRE now, before it too mutates to become more contagious.
Most people who become infected with CRE encounter the bug in hospitals or long-term-care facilities, says Lisa McGiffert, director of Consumer Reports’ Safe Patient Project.
“Our healthcare facilities are our first—and possibly our only—line of defense,” says McGiffert. “This research underscores how critically important it is for them to take concrete steps now to contain these deadly superbugs before they spread more widely.”

NOVEMBER 9, 2020
Winter is coming. With the end of Covid-19 nowhere in sight, the U.S. must brace for the annual surge of influenza, bronchitis, sinusitis, bronchiolitis, and other acute respiratory illnesses. Last winter, the Center for Disease Control and Prevention estimates there were 38 million cases of influenza alone.
One big issue is that the symptoms of these illnesses overlap considerably. A cough, runny nose, sore throat, and fever could mean the flu or other common respiratory illness. But it could also worry people that they might have Covid-19.
We are concerned that the collision of Covid-19 and winter could lead to a rash of inappropriate antibiotic prescribing. That’s a problem because, in addition to being wasteful, taking antibiotics that aren’t needed can lead to serious health issues, including allergic reactions, diarrheal infections, and the development of antibiotic-resistant bacteria that are incredibly difficult to treat.
Antibiotic resistance is such a pressing problem that the National Academies of Sciences, Engineering, and Medicine are holding a three-day workshop on the subject beginning Monday.
Inappropriate prescribing of antibiotics also drives people to get more care in the long term. More on that later.
Long before the pandemic emerged, antibiotics were greatly overused. One in three prescribed antibiotics aren’t appropriate. Acute respiratory infections are generally caused by viruses, which antibiotics don’t kill, yet acute respiratory illnesses account for 75% of all inappropriate prescriptions.
But as bacteria in the body are exposed to antibiotics, they can mutate and develop resistance to these drugs. Antimicrobial resistance causes 35,000 deaths each year in the United States alone and leads to complex diseases that have increased risk of complications and side effects. The health care costs are enormous: The CDC estimates that antibiotic resistance adds $20 billion in costs to the health care system, in addition to a $35 billion loss in productivity each year.
Several forces during the pandemic may lead to a surge in antibiotic use. SARS-CoV-2, the virus that causes Covid-19, is not affected by antibiotics. Antibiotics should be used for people with Covid-19 who also have bacterial pneumonia. Yet some physicians worry that their patients with Covid-19 might be susceptible to bacterial infections and many inappropriately prescribe antibiotics as a precaution.
The pandemic may also drive more adults and children to visit their physicians, which increases the chances they will be prescribed antibiotics. Before the pandemic, sniffles and low-grade temperatures were nothing to worry about. Now people worry that they signal the onset of Covid-19 and seek care to make sure they don’t have it. In addition, schools and day care centers have stricter rules on kids coming in who are ill.
The rapid growth of telemedicine during the pandemic may also increase the overuse of antibiotics. While telemedicine has been a lifeline for people who need care, our research has shown that, among children, telemedicine is more likely to lead to inappropriate antibiotic prescribing than in-person doctor visits. Given that a physician can’t physically examine a child remotely, he or she may not be sure about the diagnosis and prescribes antibiotics under a “better safe than sorry” approach.
While playing it safe is understandable, it represents short-term thinking and does not account for the long-term impact of antibiotics on how people seek care. Say an individual becomes ill with an acute respiratory illness, sees a physician, and is prescribed antibiotics. After a week of taking them, she feels better. Since humans are great at pattern recognition, she intuitively thinks that the antibiotics made her feel better. We know from research that she would have likely gotten better without the antibiotic, but that is not her lived experience. Naturally, the next time she gets an acute respiratory illness, she will go back to the doctor for that supposed cure.
We recently quantified this phenomenon by comparing people who were essentially randomized to low-antibiotic-prescribing physicians or to high-antibiotic-prescribing physicians. Patients who got care from high-prescribing physicians were more likely to seek care when they got sick again and received 14% more antibiotics the following year than those seeing low-prescribing physicians.
This feedforward mechanism compounds the overuse of antibiotics and self-enforces potentially inflated benefits of antibiotic use. This effect also extends across social networks as people learn from their friends and family members. We observed that spouses of people seen by high-prescribing physicians were also more likely to receive antibiotics in the subsequent year than spouses of people seen by low-prescribing physicians.
This matters in the time of coronavirus because in the months ahead, with more anxiety, more people getting sick, more people getting care, and more telemedicine use, doctors may inappropriately prescribe more antibiotics. This could dangerously accelerate the feedforward system, enforcing the misconception that antibiotics are always beneficial and driving even more inappropriate antibiotic use in the future.
Recognizing the risk of a surge of antibiotic prescribing is the first step toward encouraging physicians and patients to use antibiotics appropriately, and not for viral illnesses. It is more important than ever for physicians to take the time to talk to their patients about the appropriate use of antibiotics and reassure them that not prescribing them can be the medically right thing to do. Ideally, this will create a feedback loop such that more judicious antibiotic use in one visit may result in fewer antibiotics into the future, and help establish new norms on when antibiotics are needed.
Jowa (Zhuo) Shi is a medical student in the Harvard-MIT Program in Health Sciences and Technology. Ateev Mehrotra is an associate professor of health care policy at Harvard Medical School and a physician at Beth Israel Deaconess Medical Center.
Antibacterial ingredients in indoor dust could contribute to antibiotic resistance
Antibiotic-resistant bacteria, known as “superbugs,” pose a major public health threat. Some officials have even warned of a post-antibiotic — and sicker — era. To better understand the problem, researchers have been piecing together its contributing factors. Now in the ACS journal Environmental Science & Technology, scientists report for the first time a link between antimicrobial substances such as triclosan in indoor dust and levels of antibiotic-resistance genes, which can transfer from one bacterial cell to another.
The overuse of antibiotics in both humans and livestock has largely been blamed for the rise in drug resistance. The ubiquity of antimicrobials in hand soaps, cosmetics and other personal care products that ultimately rinse down the drain and into wastewater has also contributed. Erica Hartmann and colleagues wanted to see whether their presence in indoor dust might play a role, too.
The researchers analyzed dust samples from an indoor athletic and educational facility and found six links between antimicrobial chemicals and antibiotic-resistance genes in microbes. For instance, dust samples with higher amounts of triclosan also had higher levels of a gene that’s been implicated in bacterial resistance to multiple drugs. Although the median concentration of triclosan in indoor dust was small — much lower than amounts used in toothpaste, for example — the researchers say their findings demonstrate the need to further investigate the role of antimicrobials in dust in the rise of antibiotic resistance.
#302In the News
Antibiotic Resistance Could Take Us Back to the Days Where 40% of Us Died From Infections
HuffPost
Antibiotic Resistance Could Take Us Back To The Days Where 40% Of Us Died From Infections.
To paint a picture of a post-antibiotic apocalypse, it is necessary to understand the history.
We do not need to go back far—just to the beginning of the last century, before the discovery of our miracle drugs, antibiotics, along with preventative vaccines.
Before these truly revolutionary medical leaps, life was very different. Infections regularly killed or caused significant disabilities, many of the operations we now consider routine did not exist and the powerful drugs we use to treat cancer were unthinkable.
In particular, I want to share one key statistic—in this time before antibiotics and vaccines, around 40% of deaths were due to infections. Now, that number is just 7%.
These developments, including Sir Alexander Fleming’s discovery of the first antibiotic—penicillin—have underpinned countless medical advances since. They have allowed us to treat basic infections so surgical procedures like caesarean sections can take place safely and patients can receive chemotherapy.
People are now living longer and healthier than ever before. I think it is grossly overlooked how much of our current quality of life is down to these key achievements in medicine. Indeed antibiotics add, on average, twenty years to our lives.
But even as Sir Alexander Fleming accepted his Nobel Prize for this discovery, he foresaw a troubling future. In his acceptance speech he flagged that he was already seeing bacteria in the laboratory that developed resistance and therefore survived, and urged people to get ahead of this threat before it occurred more widely.
And here we stand, more than 70 years later, having made little progress in addressing this threat. It continues to grow globally—and this inaction seriously risks us returning to the dark ages of medicine.
Again, I do not think people fully appreciate just how much our quality of life hinges on antibiotics being effective. We are so familiar with these wonder drugs that we take them for granted. The truth is that we have been abusing antibiotics—as patients, as doctors, as travellers, as farmers and food producers globally, for short term personal gain without thinking about the future.
Many people demand them of our doctors when they simply do not need them. Surveys suggest that one in five people expect a prescription for antibiotics incorrectly believing they will treat coughs and colds caused by viruses.
Doctors have to make difficult decisions and sometimes prescribe antibiotics when they may not be needed, often because they do not have the tools to diagnose quickly and definitively. Estimates suggest that as many as half of all patients who visit their GP with a cough or cold leave with a prescription for antibiotics.
Some countries feed them to animals in huge numbers to promote growth and as a crude form of infection control. In some countries, antibiotics can be bought without prescription or online, and poor quality or fake medicines can exacerbate the problem, as well as causing harm and fatalities.
Added to that, there are many countries which we simply do not know how many antibiotics are being used and what bugs are becoming resistant, due to the lack of surveillance.
To compound these issues, we have not developed any new classes of antibiotics since the 1980s as both the public and private sector disinvested in this area and the science is challenging. Drug companies say they are too expensive to develop when they sell for such a low price.
Because of the growing threat of resistance, we are trying to limit their inappropriate use—making it far less profitable than developing drugs for other therapeutic areas such as cancer. Given the failure rate is high and it can take 10-20 years to bring an antibiotic from discovery to market, developing new antibiotics also becomes too high risk for industry due to the lack of a return on investment.
All of these failures to act are collectively driving bugs that cannot be treated by any medicines we have on this earth. Already four in 10 patients with an E.coli bloodstream infection in England cannot be treated with the commonest antibiotic (co-amoxiclav) used in hospitals. In addition, almost one in five of these bacteria were resistant to at least one of five other key antibiotics
This truly is the doomsday scenario we fear in medicine. Our apocalypse is a return to these dark ages, where people regularly died in childbirth, from stomach bugs, from simple cuts and abrasions.
The risk is worrying enough that the UK Government put this issue on its national risk register—up there with security threats, floods and pandemic flu.
And already we are seeing horror cases worthy of this classification. In the United States earlier this year, we saw a tragic case where a woman died after contracting an infection that we simply did not have the medicine to treat.
All 14 kinds of antibiotics the hospital had on hand did not work. Even more stark—none of the 26 antibiotics available in the United States would have worked either.
RELATED…
I Almost Lost My Arm Because Of Antibiotic Resistance
http://www.huffingtonpost.co.uk/ent…biotic-resistance_uk_5a54eecce4b01e1a4b1a15c3
Yet, this is just one example and it is important to stress that this is not just a future threat—it is happening now. Already, it is estimated that 700,000 people across the world die from drug-resistant infections.
And indeed the full scale of potential future doom has been forecast. Lord O’Neill’s Independent Review on this topic predicted that without action, by 2050 the world could see 10 million people dying per year from drug resistant infections, at a cost of more than $100 trillion to the global economy. That’s one person every three seconds. It is also predicted to push an additional 28.3 million people into extreme poverty.
Now you know how bad it could get—so what are we doing about it you might ask?
Thankfully, the answer is quite a lot.
I have made it a personal mission to bring this issue to the attention of the world, and we are beginning to see progress.
In 2016 we brought this issue to the United Nations and secured a landmark declaration from all 193 countries. They all pledged to take action to curb this threat. I am now part of a global group driving forward this work.
We have kept it high on the political agenda though Germany’s G20 Presidency and most recently through a declaration at the UN Environment Assembly—the world’s highest-level decision-making body on the environment. This now commits the 193 UN Member States to not only take action in humans and animals but also to protect our environment and address the contribution the environment has to play on the emergence and spread of drug resistance.
This issue has the attention of some of our brightest minds in academic and the private sector, and important charitable organisations like the Bill and Melinda Gates Foundation and Wellcome Trust.
I am also very proud that the UK is truly leading the way, spending over £615 million on research, awareness raising work, and supporting developing countries. This includes a £265 million investment to support these countries to develop and improve their surveillance of antibiotic use and spread of resistance. Our human and animal health professionals have made huge strides in reducing our use and sales of antibiotics and have committed to do more.
While global progress is far too slow for my liking, this work is good news and its complex nature means it is a long-term problem that we must find sustainable solutions to. But the good news is that we can all do our bit to halt the rise of drug-resistant infections.
Simple things like good hygiene and hand-washing stop the spread of infections. Even better, get the vaccines that are offered to you. And also please do not ask your healthcare professionals for antibiotics—they will know best, so please listen to them and follow their instructions when they do prescribe. It is vital they are there and used appropriately when you or a loved one needs them.
While this post-antibiotic apocalypse looks dark, there is certainly hope. But much more needs to be done at all levels and across all sectors to avoid passing this doomsday scenario on to our children and grandchildren.
HuffPost UK Tech has launched HuffPost-
Apocalypse, a project that aims to investigate what an apocalypse would mean for humanity, how we can best delay the end of the world, what the world will look like after we’re gone and what the best viable options for survival will be for anyone left. Join in the conversation with #HuffPostApocalypse on Twitter. To read more from the series, visit our dedicated page.
..But even as Sir Alexander Fleming accepted his Nobel Prize for this discovery, he foresaw a troubling future. In his acceptance speech he flagged that he was already seeing bacteria in the laboratory that developed resistance and therefore survived, and urged people to get ahead of this threat before it occurred more widely.
And here we stand, more than 70 years later, having made little progress in addressing this threat. It continues to grow globally—and this inaction seriously risks us returning to the dark ages of medicine..
All of these failures to act are collectively driving bugs that cannot be treated by any medicines we have on this earth. Already four in 10 patients with an E.coli bloodstream infection in England cannot be treated with the commonest antibiotic (co-amoxiclav) used in hospitals. In addition, almost one in five of these bacteria were resistant to at least one of five other key antibiotics
And yet Big Pharma and .Gov have information that simply adding some EIS/CS with an antibiotic multiplies the effectiveness of the antibiotic, ALL antibiotics.
Just no new Revenue Stream is developed, meaning no new exorbitant price gouging can be created. Big Pharma is Snake-oil Salesmen.